Abstract¶
This study explores the effects of enzyme concentration and pH on the activity of turnip peroxidase, an enzyme involved in plant defense and oxidative stress regulation. The experiment tested varying enzyme concentrations and pH levels, using hydrogen peroxide as the substrate, to measure reaction rates. The results revealed that increasing enzyme concentration led to a modest increase in reaction rate, with a plateau observed due to substrate saturation. pH variation showed optimal activity near neutral conditions (pH 6.5–7.0), while extreme acidic and basic environments resulted in decreased enzyme efficiency. These findings highlight the importance of enzyme optimization for applications in agriculture and industry, where controlling enzyme conditions is critical for process efficiency. Further studies should explore the impact of other variables such as temperature and substrate concentration to enhance the understanding of peroxidase activity..
Introduction¶
Enzymes are biological catalysts that accelerate chemical reactions in living organisms by lowering the activation energy required for reactions to occur, playing essential roles in processes such as metabolism, digestion, and cellular signaling (Kontro & Oja (1981)). Peroxidases, a diverse group of enzymes, catalyze reactions involving the oxidation of substrates using hydrogen peroxide as an electron acceptor. Turnip peroxidase, derived from Brassica rapa, plays a significant role in plant defense and development, catalyzing reactions crucial for lignin biosynthesis and reactive oxygen species metabolism (Belinky et al. (2003)).
The chemical reactions involved in turnip peroxidase activity are as follows:
Reduction of Hydrogen Peroxide (H₂O₂): Peroxidase facilitates the decomposition of H₂O₂, reducing it into water:
Oxidation of Substrate: The reaction regenerates the active enzyme and oxidized substrate.
Understanding the effects of enzyme concentration and pH on the activity of turnip peroxidase is critical for optimizing its applications in agriculture and industry, such as bioremediation, food processing, and biosensors. This experiment hypothesized that an increase in enzyme concentration would elevate reaction rates and that enzymatic activity would peak at an optimal pH, declining under acidic or basic conditions.
Methods¶
To investigate the effects of enzyme concentration and pH on turnip peroxidase activity, reaction mixtures were prepared with a fixed substrate concentration of hydrogen peroxide, a phosphate buffer, and varying conditions for enzyme concentration and pH.
Enzyme Extraction and Preparation Turnip peroxidase was extracted by blending 50 grams of turnip tissue in 200 mL of cold phosphate buffer (pH 7.0) for 2 minutes. The homogenate was filtered through cheesecloth, and the filtrate was centrifuged at 4,000 rpm for 15 minutes. The resulting supernatant was collected as the enzyme extract and stored on ice until use.
Reaction Setup Three experimental groups were established:
- Control Group: Reaction mixtures contained all components except the enzyme to ensure that any observed changes in absorbance were due to enzymatic activity.
- Enzyme Concentration Group: Reaction mixtures were prepared with increasing volumes of enzyme extract (e.g., 0.5 mL, 1.0 mL, and 1.5 mL) to test the effect of enzyme concentration on reaction rates.
- pH Variation Group: Reaction mixtures included buffer solutions adjusted to pH 4.0, 7.0, and 9.0 using HCl or NaOH to determine the effect of pH on enzyme activity.
Each reaction mixture consisted of 1.5 mL of phosphate buffer, 0.5 mL of hydrogen peroxide (0.1 M), and the appropriate volume of enzyme extract or pH-adjusted buffer. The total reaction volume was maintained at 3.0 mL. Measurement of Enzyme Activity The reaction mixtures were transferred to cuvettes, and absorbance was measured at 470 nm using a spectrophotometer. The initial absorbance was recorded at time zero, and subsequent measurements were taken every 30 seconds for a total of 5 minutes. Each experimental condition was tested in triplicate to ensure accuracy and reproducibility. The control group lacked enzyme extract to serve as a baseline for comparison..
Results¶
The control group showed constant absorbance values, confirming the absence of enzymatic reactions. In the increased enzyme concentration group, the absorbance increased over time, indicating enhanced enzymatic activity. However, the rate of increase plateaued after 120 seconds, suggesting substrate saturation. In the pH variation group, enzymatic activity was highest at pH 6.5–7.0, with noticeable declines at pH levels below 5 or above 8. This indicates that extreme pH conditions disrupt enzyme activity, likely due to denaturation or alterations in the enzyme’s active site.
A line graph was generated to depict absorbance changes over time for all experimental groups.
| Time (s) | Control | Increased Enzyme Concentration | HCl Added |
|---|---|---|---|
| 0 | 0.138 | 0.142 | -0.017 |
| 30 | 0.697 | 0.605 | 0.011 |
| 60 | 0.698 | 0.665 | 0.01 |
| 90 | 0.695 | 0.668 | 0.009 |
| 120 | 0.697 | 0.67 | 0.01 |
| 150 | 0.694 | 0.666 | 0.009 |
| 180 | 0.695 | 0.667 | 0.009 |
| 210 | 0.696 | 0.669 | 0.01 |
| 240 | 0.694 | 0.67 | 0.008 |
| 270 | 0.639 | 0.668 | 0.01 |
| 300 | 0.644 | 0.667 | 0.01 |
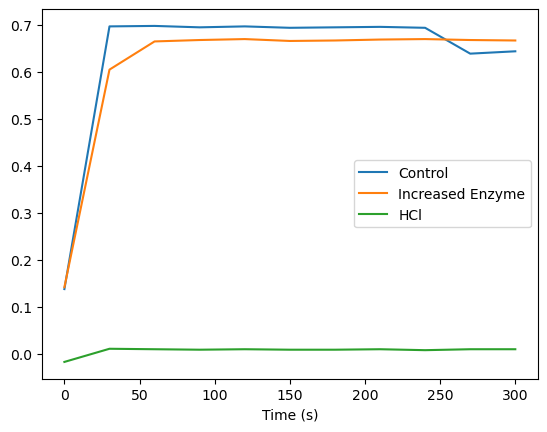
Figure 1:This reflects the effects of different variables on enzymatic activity. The activity flucates per each variable as time goes on after one of the variables are added.
Figure 1 illustrated that increasing enzyme concentration initially accelerated reaction rates until substrate availability became limiting. Similarly, the pH variation data showed a clear peak in enzymatic activity at near-neutral pH, with steep declines in acidic and basic conditions.
Discussion¶
The results demonstrated a positive correlation between enzyme concentration and reaction rate, as hypothesized. The increased absorbance observed with higher enzyme concentrations reflects the principles of Michaelis-Menten kinetics (Schnell (2000)), where the rate of reaction rises with enzyme availability until substrate saturation is reached. However, the anticipated linear relationship was not entirely observed, likely due to substrate limitations or experimental variability in mixing and measurement.
The pH-dependent activity showed a distinct optimal range around neutrality, with enzymatic activity diminishing significantly in acidic or basic environments. This can be attributed to alterations in the enzyme’s tertiary and quaternary structure, which impact the active site conformation and substrate binding. Extreme pH levels may lead to denaturation, rendering the enzyme inactive. These findings align with prior studies on peroxidases, which underscore the importance of maintaining a stable pH environment to ensure enzyme functionality.
The results highlight the practical implications of optimizing enzyme concentration and pH for specific industrial processes. For example, in agricultural applications, peroxidase-based biosensors for detecting environmental pollutants or oxidative stress in plants would require precise control of these factors to ensure reliability and efficiency.
Limitations of this study include the relatively narrow pH range tested and potential inconsistencies in enzyme preparation and substrate concentration. Future experiments should focus on expanding the pH range, exploring other environmental variables like temperature, and employing more refined measurement techniques to improve data accuracy and reproducibility.
Conclusion¶
This study confirmed the catalytic activity of turnip peroxidase and demonstrated how enzyme concentration and pH influence reaction rates. The positive correlation between enzyme concentration and reaction rate underscores the enzyme’s efficiency, while the pH findings emphasize the need to maintain near-neutral conditions for optimal activity. These insights are critical for leveraging turnip peroxidase in agricultural and industrial applications, where enzyme optimization can significantly enhance process efficiency and reliability.
Future research should investigate the effects of temperature, substrate variability, and broader pH ranges to further understand the behavior of peroxidase under diverse conditions, laying the groundwork for novel applications in biosensing and biocatalysis.
Data Availability¶
The data that support the findings fo this study are openlly availible in [jterm-2025] at https://
- Kontro, P., & Oja, S. S. (1981). Enzyme Kinetics. In Methods in Neurobiology (pp. 265–337). Springer US. 10.1007/978-1-4684-3806-2_5
- Belinky, P. A., Flikshtein, N., Lechenko, S., Gepstein, S., & Dosoretz, C. G. (2003). Reactive Oxygen Species and Induction of Lignin Peroxidase in Phanerochaete chrysosporium. Applied and Environmental Microbiology, 69(11), 6500–6506. 10.1128/aem.69.11.6500-6506.2003
- Schnell, S. (2000). Enzyme Kinetics at High Enzyme Concentration. Bulletin of Mathematical Biology, 62(3), 483–499. 10.1006/bulm.1999.0163